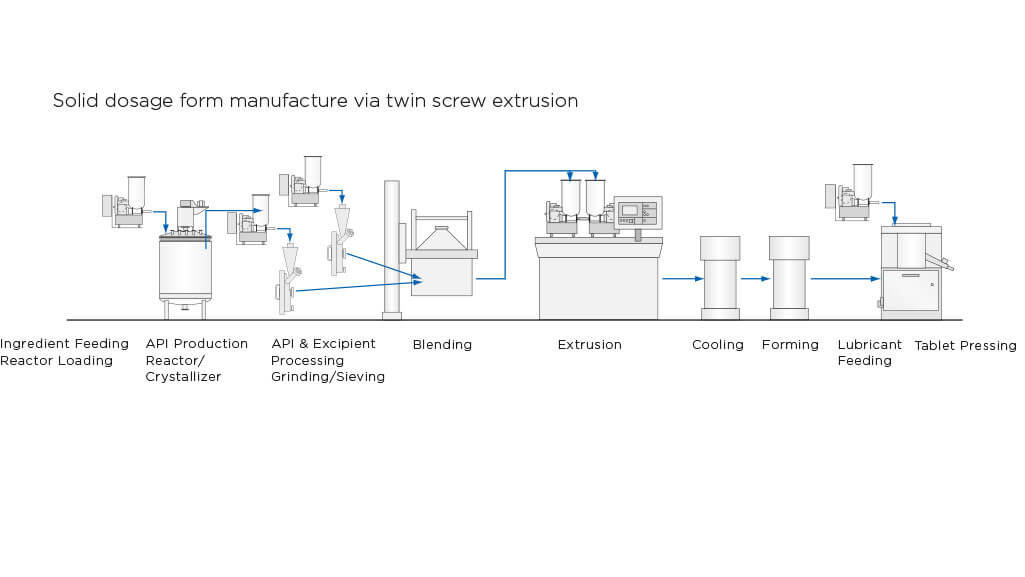
Solid dosage form manufacture via twin screw extrusion
The application of continuous extrusion via twin screw extruders for pharmaceutical solid dosage form production is growing in popularity as a preferred option to more traditional granulation techniques. Coperion's twin screw extruders offers several advantages including continuous processing with reproducibility and combination of a variety of unit operations within one piece of equipment, i.e. distributive and dispersive mixing, granulating and devolatilization. The production method provides gentle product handling at short residence time and low temperature.
Two methods in which the Coperion ZSK extruder is typically used for solid dosage form production are hot melt extrusion and wet granulation via wet extrusion. Coperion is one of the pioneers in the development of pharmaceutical extrusion. Coperion has references for pharmaceutical extruders in the sizes ZSK 18 to ZSK 70. The excellent mixing behavior and the devolailization possibilities of the co-rotating twin screws make this system particularly suitable for pharmaceutical extrusion.
During hot melt extrusion and wet extrusion powders and liquids must be fed accurately and continuously in order to ensure consistency of formulation, constant throughput, proper order of mixing ingredients and regulated mass transfer. Coperion K-Tron Pharmaceutical Loss-in-weight (LIW) feeders are used in conjunction with extruders in the pharmaceutical industry for both powder and liquid feeding. Typically, feed streams are introduced to the extruder in a “starve-fed” manner, where the rate is set by the feeders and is independent of the extruder screw speed.
Typical raw materials:
- APIs of various potencies
- Polymers including polyethylene glycol, polyethylene oxide, hydroxypropylcellulose, ethyl cellulose, hydroxymethyl cellulose (HPMC), polymethylacrylate, polyvinylpyrrolidone, polyvinylacetate, Povidone