Hygienic Design Feeders, Components and Conveying Systems for Sanitary Applications
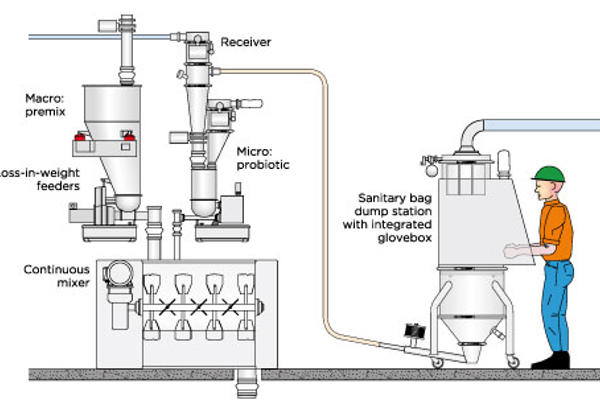
The production of dry dairy powders and infant/baby formulas requires extremely sanitary design of the equipment as well as high accuracy in the precise addition of ingredients to the formula blend. The system provided must also offer the versatility to handle a wide variety of difficult flowing and temperature-sensitive ingredients, including vitamins, nutrient additives and even probiotics. In addition, depending upon the type of process and the processing environment, the equipment must be able to be cleaned by either wet or dry cleaning methods. The hygienic designs provided by Coperion and Coperion K-Tron are easily adaptable for all of these requirements.
Products and ingredients may include: milk powder, whey powder, lactose, casein, cheese powder, infant formula, maltodextrine, probiotics, vitamins, baby food
 Coperion
Open navigation
Coperion
Open navigation